RUES3
Click here for a list of transgenic lines available from the Brivanlou Laboratory
RUES3 (NIH Registration number 0042)
Biosafety Level: 2
Shipped: Frozen
Medium & Serum / Propogation / Subculturing / Preservation: See Protocols
Organism: Homo sapiens (human)
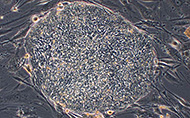
Morphology: Spherical colony
Cell Type: Human embryonic stem cell
Permits/Forms: Completion of the implementing letter for the UBMTA between the receiving institution and The Rockefeller University. In addition to the implementing letter mentioned above, other regulatory permits may be required for the transfer of these materials, e.g., import or export permits for transfers to non-US recipients. The requesting institution is responsible for obtaining the permits.
Restrictions: Cannot be used for diagnostic or therapeutic use; for use in teaching and non-commercial research only.
Cytogenetic Analysis: XY
Age: embryo, blastocyst
Comments: RUES3 is a human embryonic stem cell line derived from a de-identified frozen embryo that was originally generated for reproductive purposes and later donated with informed consent for research purposes. The Provider does not have any information that could be used to identify the embryo and/or gamete donors. RUES3 cells are pluripotent and can differentiate to representatives of the three primary germ layers.
References: Recipients are requested to provide notice of publications citing use of this line to the Provider.
Notices and Disclaimers
The Material is experimental in nature and must be used with prudence and appropriate caution, since not all of its characteristics are known. The Material is provided without warranty of merchantability or fitness for a particular purpose or any other warranty, express or implied. Provider makes no representation or warranty that the use of the Material will not infringe any patent or other proprietary right. The Material cannot be used for any commercial purpose or for work involving human subjects, including diagnostic testing.